
"Source of power" that changes the future of the automobile

Invisible power that has contributed to the improvement of the performance of TOYOTA MIRAI, the world first mass production fuel cell vehicle. Chemical reaction of the electrode catalyst will contribute to further dissemination of the fuel cell vehicle.
HOME > Automotive Engineering Exposition 2017 > Electrode catalyst for fuel cell
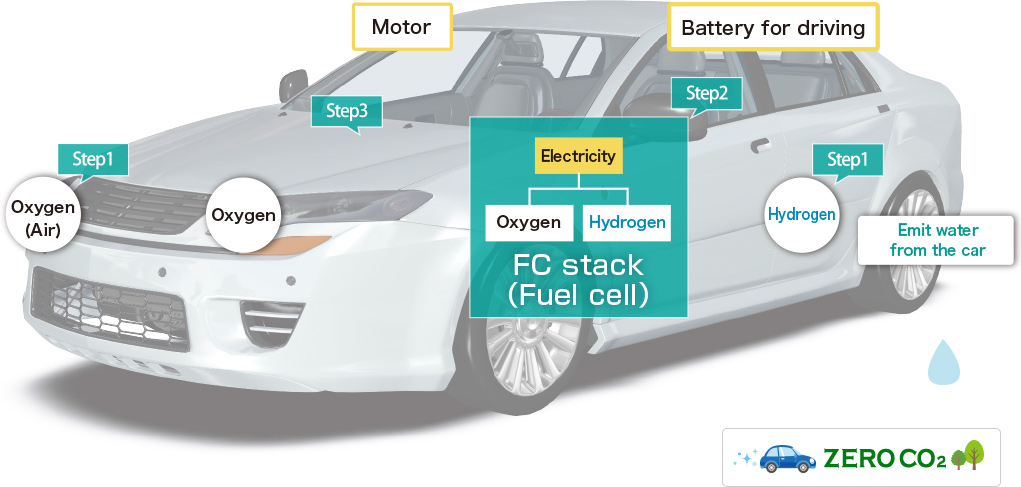
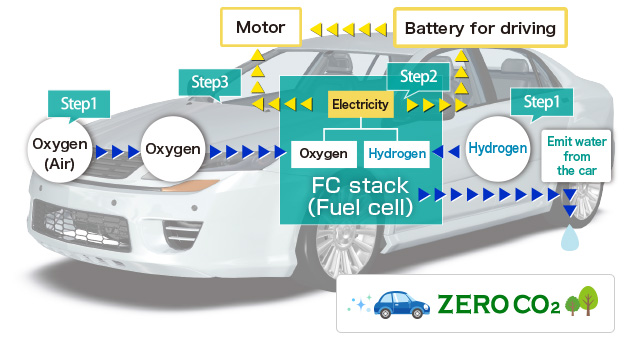
Fuel cell is the "Electrical generator" which can generate electrical energy continuously by electrochemical reaction between oxygen and hydrogen. It is different from the primary battery such as dry cell and rechargeable battery that needs to repeat charging. We can utilize electrical energy that the fuel cell generates through electrochemical reaction between hydrogen as fuel and oxygen. In fact, the fuel cell vehicle is a car that is equipped with "Fuel cell (Stack)" which generates electricity through chemical reaction between hydrogen and oxygen to power a motor. Hydrogen is an environment-friendly and alternative energy to gasoline, that can be produced from a variety of raw materials.


This car works by motor power with generates electricity to power a motor through the chemical reaction between hydrogen and oxygen. It's different from gasoline engine car that burns gasoline. Fuel cell vehicle is the ultimate eco-friendly car since it doesn’t burn gasoline(fossil fuels) and doesn't emit CO2(Carbon dioxide) which is a cause of global warming. In addition, it is an environmentally-friendly car without noise as it doesn't have an engine.


Electrode catalyst is used in a electrode(※) of the Solid Polymer Fuel Cell. Fuel cell produces water and electricity by reaction between hydrogen and oxygen which is opposite mechanism of producing hydrogen and oxygen by electrolysis of water. Electrode catalyst supports reaction between hydrogen and oxygen in a fuel cell.
Electrode is a conductor that is on anode, the positive terminal for current flow out and on cathode, the negative terminal for current flow in. It is an electrical contact part for the purpose of moving target object or measuring electric signal.
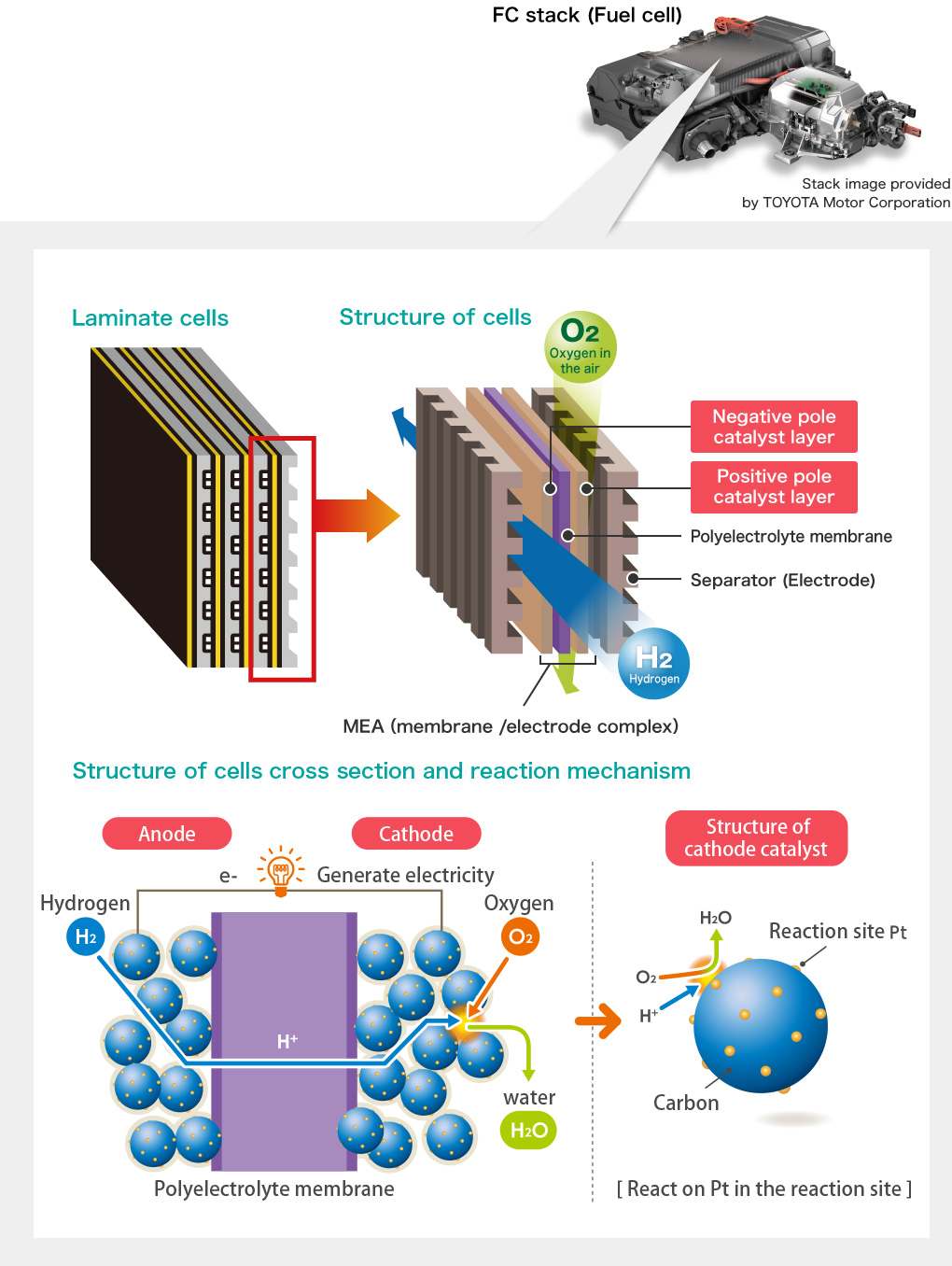
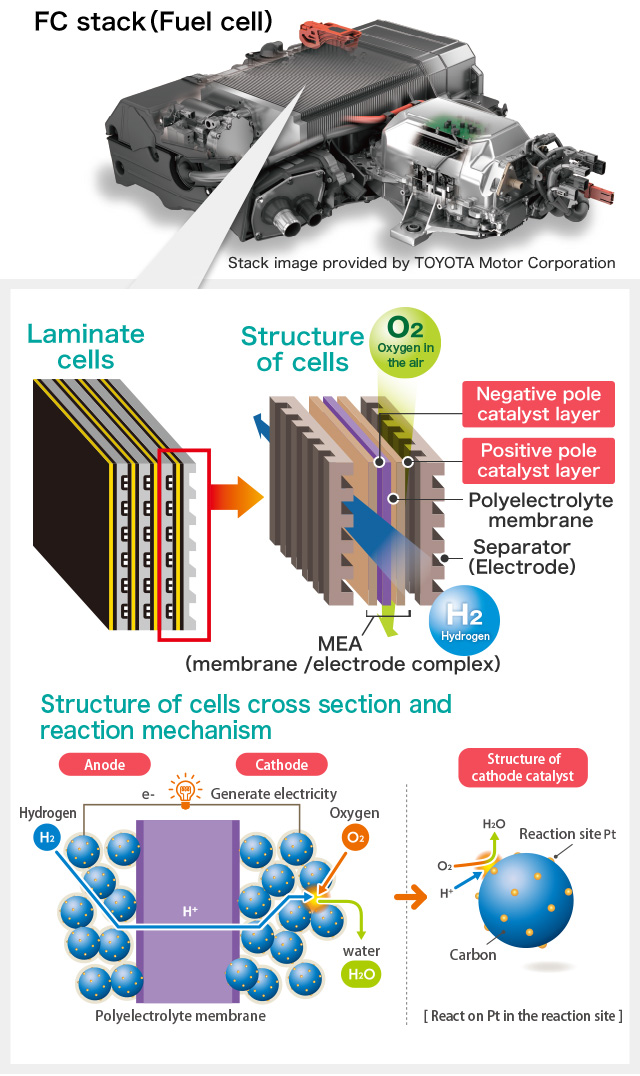
The part where generates electricity in the fuel cell is called "Stack" which consists of MEA(※) aggregation between separators(※). Gas flow path for hydrogen(fuel) and oxygen(air) is formed in several separators. Hydrogen enters at the fuel electrode (positive terminal/anode) and oxygen enters at the air electrode (negative terminal/cathode) of MEA and generate electricity by the electrochemical reaction.
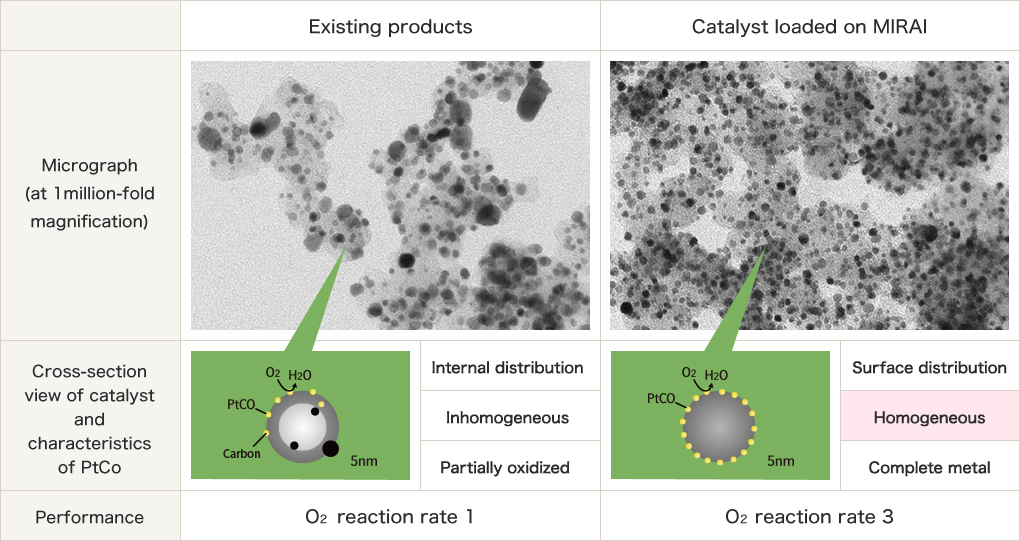
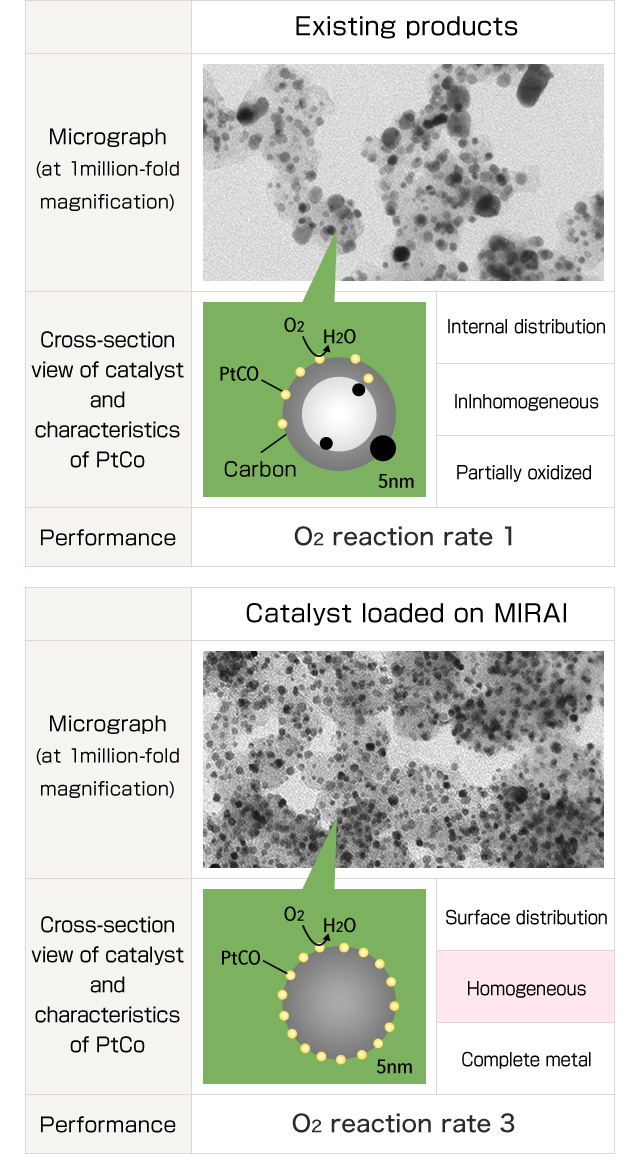
Electrode catalyst is the key material to affect improvement of output of the fuel cell vehicle and fuel efficiency of H2. The most-advanced technique of electrode catalyst jointly developed by TOYOTA Motor Corporation and us was adopted to their world's first FC vehicle on mass production.


![Example of developed technology [Pt uniformly loading technology]](../images/fc/fig04.jpg)
![Example of developed technology [Pt uniformly loading technology]](../sp/images/fc/fig04.jpg)


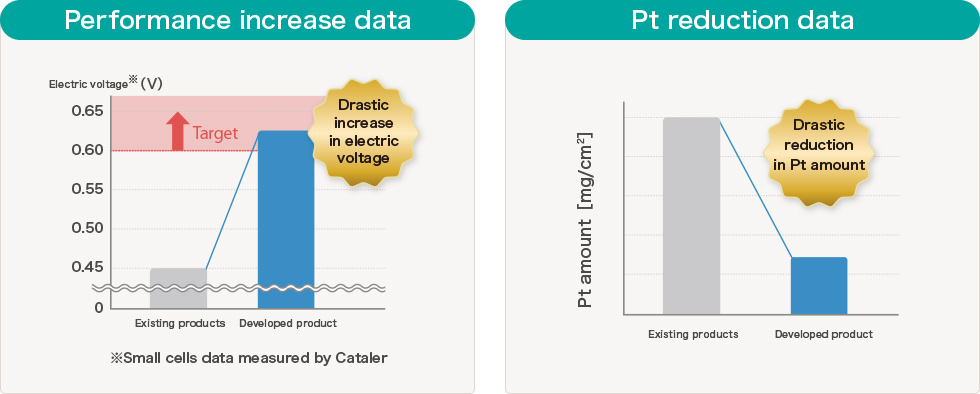
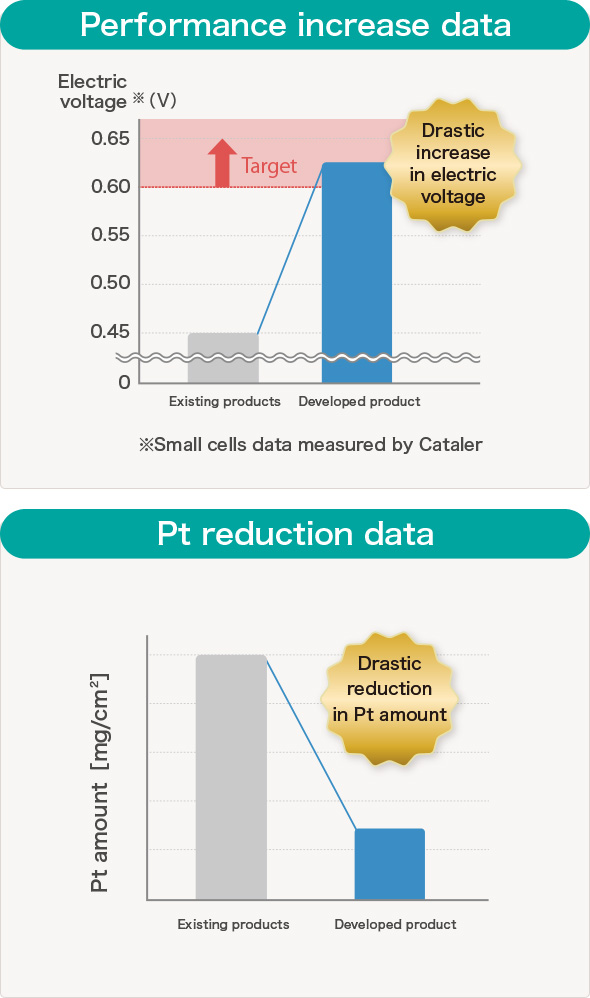
Our developed "Electrode catalyst for Fuel Cell (that's been installed on the Fuel Cell vehicle MIRAI) has achieved cost-reduction due to significant reduction in use of expensive Platinum and contributed to improve reliability of the FCV on the series-production as well as contributing to improvement of performance of MIRAI by improving fuel cell output, H2 fuel efficient, and high temperature output when driving upslope that were the challenges.


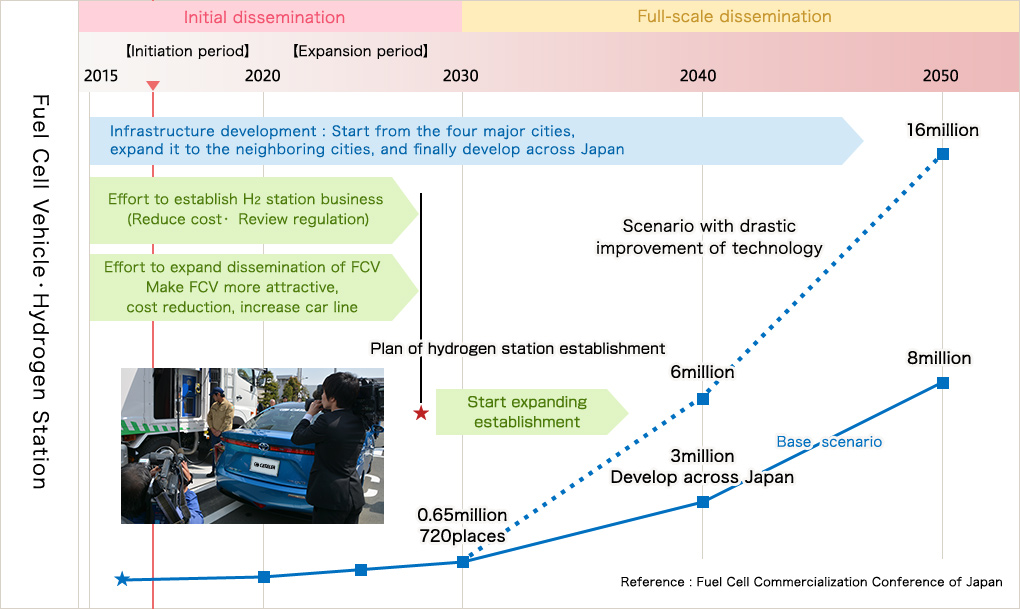
FCCJ, Fuel Cell Commercialization Conference of Japan, that makes policy proposals for solving the problem in practical use and dissemination of the fuel cell in Japan, established start time of FCV dissemination to the general users in 2015 as a mile stone. And they announced their scenario of "Aim to start dissemination of FCV to the general users from 2015 and establish an infrastructure for hydrogen supply to ensure user-friendliness" with agreement of the major automobile companies and domestic energy companies. Hydrogen ・Fuel cell Strategy Council of Ministry of Economy, Trade and Industry is planning a roadmap for FCV dissemination and hydrogen station improvement in short term for 2030.